CHRONOLOGY MODULE
Vocabulary
Absolute Dating:
The process of determining when an event occurred along a calendrical timeline.
Biosphere:
The component of the Earth system which consists of all life on earth, including all avian, terrestrial, and aquatic species. The biosphere can be understood in terms of the abundance of living organisms on earth (global biomass) and in terms of its internal organization or systemic process (ecosystems).
Carbon-12 (12C):
The most abundant stable carbon isotope occurring in nature. 12C containes 6 protons and 6 neutrons.
Carbon-13 (13C):
A naturally occurring, stable carbon isotope, which is considerably less abundant in the atmosphere than is 12C but considerably more abundant than 14C. 13C contains 6 protons and 7 neutrons.
Carbon Cycle:
The process by which carbon fl ows throughout and is exchanged between various physical and biological systems (for example, the atmosphere, the oceans, and the biosphere). Because carbon is an essential ingredient for life on earth, it is critical that carbon be continuously available to organisms, so understanding how carbon is recycled through the carbon cycle is of central importance for biologists.
Carbon dioxide (CO2):
A molecule consisting of two oxygen atoms and one carbon atom. Carbon dioxide molecules form in the atmosphere, and their carbon atoms can be either 12C, 13C, or 14C atoms.
Dated event:
An event which is directly dated by a particular dating method. In the case of radiocarbon dating, the dated event is the time of death of an organism from which a sample is taken. The dated event may or may not be of direct interest to archaeological research.
Isotope:
A variant form of an element. Diff erent isotopes of the same element have diff erent numbers of neutrons in their atomic nuclei. For example, a 12C atom has six neutrons in its nucleus, 13C has seven, and 14C has eight, yet all three are still carbon atoms and interact in the same chemical reactions in the same way.
Law of superposition:
A principle of geology which states that, in a sequence of geological layers, a lower layer of sediments was deposited before, and therefore is older than, overlying layers. This law only applies in cases where such layers have not been disturbed or mixed since the time of their deposition.
Neutron:
A subatomic particle which has no charge. Together with protons, neutrons are one of the building blocks of the nuclei of atoms, but they can also occur free in nature.
Nitrogen-14 (14N):
A common, naturally occurring, stable nitrogen isotope. 14N contains 7 protons and 7 neutrons.
Radiocarbon (14C):
A naturally occurring, radioactive carbon isotope, which is considerably less abundant than both 12C
and 13C.
Relative dating:
The process of determining whether an event came before or after another event in time, without consideration for how much time intervened between the two or how long ago in the past they occurred.
Stratigraphy:
A sequence of layers at an archaeological site or a geological locale that represents the depositional history of that location.
Stratum (plural: strata):
A layer in a geological deposit having characteristics (age, color, composition) that make it distinguishable from other layers.
Target event:
An archaeological term used to refer to the event which is of interest to an archaeologist and for which they would like to estimate a date. Linking a particular "dated event" to the "target event" is one of the biggest challenges of historical sciences like archaeology and geology.
Years BP (years before present):
Scientists who use radiocarbon dating agreed to always report the radiocarbon dates using 1950 as the point of origin, the point from which time is counted back. So, a radiocarbon date which is presented as "537 years BP" means 537 years before present. In other words, it is 1413 AD.
The process of determining when an event occurred along a calendrical timeline.
Biosphere:
The component of the Earth system which consists of all life on earth, including all avian, terrestrial, and aquatic species. The biosphere can be understood in terms of the abundance of living organisms on earth (global biomass) and in terms of its internal organization or systemic process (ecosystems).
Carbon-12 (12C):
The most abundant stable carbon isotope occurring in nature. 12C containes 6 protons and 6 neutrons.
Carbon-13 (13C):
A naturally occurring, stable carbon isotope, which is considerably less abundant in the atmosphere than is 12C but considerably more abundant than 14C. 13C contains 6 protons and 7 neutrons.
Carbon Cycle:
The process by which carbon fl ows throughout and is exchanged between various physical and biological systems (for example, the atmosphere, the oceans, and the biosphere). Because carbon is an essential ingredient for life on earth, it is critical that carbon be continuously available to organisms, so understanding how carbon is recycled through the carbon cycle is of central importance for biologists.
Carbon dioxide (CO2):
A molecule consisting of two oxygen atoms and one carbon atom. Carbon dioxide molecules form in the atmosphere, and their carbon atoms can be either 12C, 13C, or 14C atoms.
Dated event:
An event which is directly dated by a particular dating method. In the case of radiocarbon dating, the dated event is the time of death of an organism from which a sample is taken. The dated event may or may not be of direct interest to archaeological research.
Isotope:
A variant form of an element. Diff erent isotopes of the same element have diff erent numbers of neutrons in their atomic nuclei. For example, a 12C atom has six neutrons in its nucleus, 13C has seven, and 14C has eight, yet all three are still carbon atoms and interact in the same chemical reactions in the same way.
Law of superposition:
A principle of geology which states that, in a sequence of geological layers, a lower layer of sediments was deposited before, and therefore is older than, overlying layers. This law only applies in cases where such layers have not been disturbed or mixed since the time of their deposition.
Neutron:
A subatomic particle which has no charge. Together with protons, neutrons are one of the building blocks of the nuclei of atoms, but they can also occur free in nature.
Nitrogen-14 (14N):
A common, naturally occurring, stable nitrogen isotope. 14N contains 7 protons and 7 neutrons.
Radiocarbon (14C):
A naturally occurring, radioactive carbon isotope, which is considerably less abundant than both 12C
and 13C.
Relative dating:
The process of determining whether an event came before or after another event in time, without consideration for how much time intervened between the two or how long ago in the past they occurred.
Stratigraphy:
A sequence of layers at an archaeological site or a geological locale that represents the depositional history of that location.
Stratum (plural: strata):
A layer in a geological deposit having characteristics (age, color, composition) that make it distinguishable from other layers.
Target event:
An archaeological term used to refer to the event which is of interest to an archaeologist and for which they would like to estimate a date. Linking a particular "dated event" to the "target event" is one of the biggest challenges of historical sciences like archaeology and geology.
Years BP (years before present):
Scientists who use radiocarbon dating agreed to always report the radiocarbon dates using 1950 as the point of origin, the point from which time is counted back. So, a radiocarbon date which is presented as "537 years BP" means 537 years before present. In other words, it is 1413 AD.
Background Information
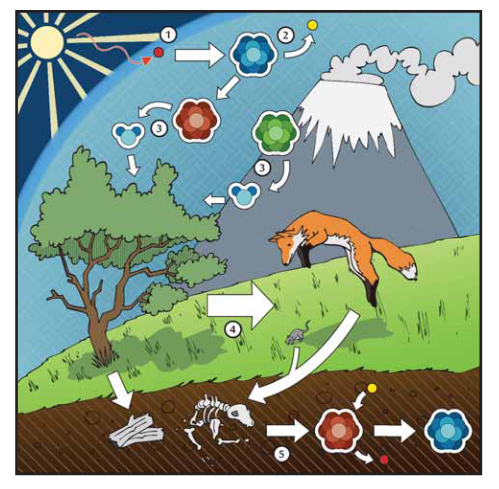
When cosmic rays excite atmospheric neutrons, some of these neurons collide with atmospheric nitrogen-14 (14N), which is transformed into radiocarbon (14C) as a result (see Appendix B). This 14C constitutes a miniscule proportion of atmospheric carbon, alongside two considerably more abundant carbon isotopes: carbon-12 (12C) and carbon-13 (13C). All three of these carbon isotopes combine with atmospheric oxygen atoms to form carbon dioxide molecules. Through the process of photosynthesis, plants incorporate carbon from these carbon dioxide molecules into their tissues, maintaining a 14C to 12C ratio in equilibrium with the atmosphere as long as they are alive. In turn, animals eat plants or other animals, and the carbon in the plant or animal tissues that they consume is incorporated into their own tissues. When plants and animals die, they cease incorporating new carbon into their tissues and the "radiocarbon clock" starts ticking.
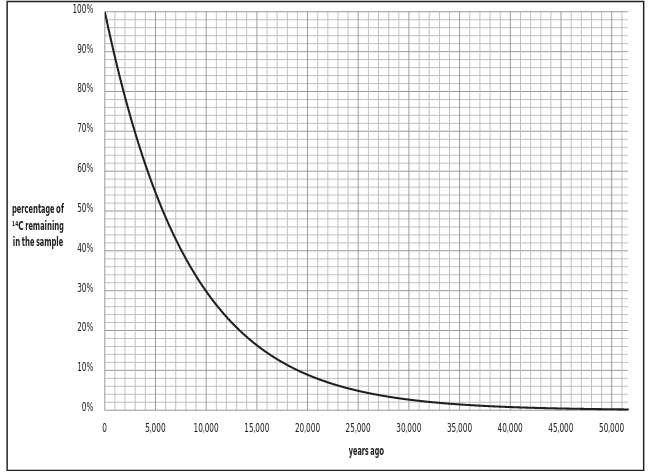
Radiocarbon is a radioactive isotope which decays back into 14N at a constant rate: after approximately 5,700 years, half of the amount of 14C which was originally in the sample converts back into 14N. After another ~5,700 years, half of the remaining 14C converts into 14N. This process of radioactive decay continues indefinitely through time, but the amount of 14C remaining in a sample becomes so small after approximately 50,000 years that laboratory machines have a hard time detecting it. Conversely, 12C is a stable isotope, so the amount of 12C that is present in a sample at the time of its death should remain constant over time.
Technicians who work at radiocarbon laboratories measure the amount of 14C and 12C remaining in a sample of organic material (such as wood, charcoal, bone collagen, shell, hair, seeds, or plant fibers). If they assume that the ratio of 14C to 12C that was originally present in the sample is identical to the ratio of modern atmospheric 14C to 12C, then they can use their measurement of the amount remaining in a sample to estimate the amount of time that has passed since the death of the organism that the sample came from, assuming a constant rate of radiocarbon decay.
If an archaeologist or geologist has good reason to believe that the death of a sample (the “dated event”) corresponds closely in time with its deposition at an archaeological or geological site (the “target event”), they can use this sample’s date to determine when it was deposited at the site, allowing them to begin to construct a timeline for the archaeological or geological history of that site
C14 in the Carbon Cycle
Describe in your own words the five steps in the radiocarbon formation and decay.
1. What is the approximate age of an organic sample with 75% radiocarbon
remaining?
2. How much radiocarbon remains in a sample after 3 half-lives?
3. What is the approximate age of an organic sample with 1/512 of the original
radiocarbon remaining?
4. How much radiocarbon remains in a sample after 25,000 years?
remaining?
2. How much radiocarbon remains in a sample after 3 half-lives?
3. What is the approximate age of an organic sample with 1/512 of the original
radiocarbon remaining?
4. How much radiocarbon remains in a sample after 25,000 years?
Part 1
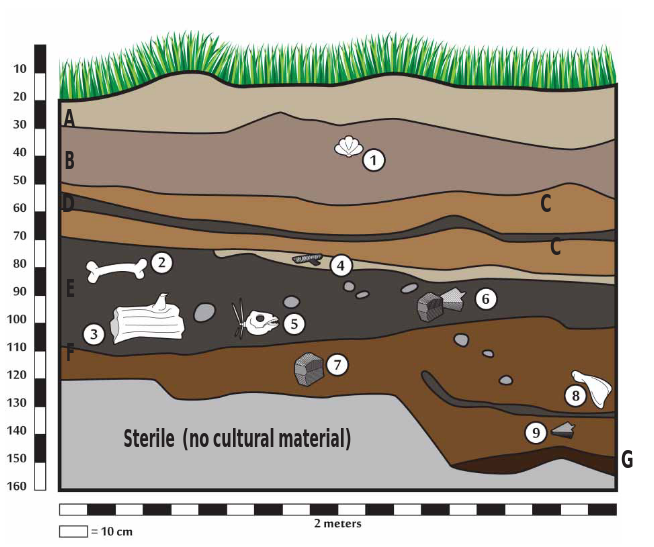
You are the head of an archaeology crew. You and your crew just returned from an excavation of a Test pit (Test Pit 1) at the Rasshua 1 site on Rasshua Island in the Kuril Island chain. After the excavation, your fi eld assistants drew the stratigraphic
column of one of the walls of the excavation. Using the drawing of the stratigraphy, you now have to answer the following questions in order to decide how you are going to establish the chronology, or the sequence of events, of this site.
column of one of the walls of the excavation. Using the drawing of the stratigraphy, you now have to answer the following questions in order to decide how you are going to establish the chronology, or the sequence of events, of this site.
1. Which layers can you date using 14C? Why do you think so?
2. What materials would you date to fi nd out the age of cultural occupations? Why?
3. What would you date to fi nd out the age of layer B?
4. What materials would you date to fi nd out the age of Ushishir tephra (Layer C)?
5. How would you test the idea that volcanic eruptions had a devastating consequence for human occupation?
2. What materials would you date to fi nd out the age of cultural occupations? Why?
3. What would you date to fi nd out the age of layer B?
4. What materials would you date to fi nd out the age of Ushishir tephra (Layer C)?
5. How would you test the idea that volcanic eruptions had a devastating consequence for human occupation?
Part 2
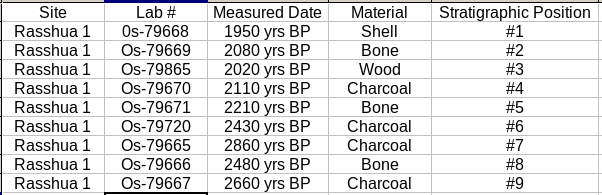
After you answered these questions and decided which levels you wanted to date you sent your radiocarbon samples to a lab that processes the samples and determines the date of each sample. Take a look at the results that they sent back. Now answer the following questions to interpret what events took place at this site and how they are related to each other in time.
1. Are there any out of sequence dates? How would you explain them?
(Hint: Think back to target and dated events and to what human or natural activities can disturb certain levels)
2. How long did humans occupy the site? Were there any gaps in occupation?
3. What is the age of artifact X? How did you determine it? What are the dated and target events for this sample?
(Hint: Think back to target and dated events and to what human or natural activities can disturb certain levels)
2. How long did humans occupy the site? Were there any gaps in occupation?
3. What is the age of artifact X? How did you determine it? What are the dated and target events for this sample?
Part 3
The last step in the process of archaeological analysis of a site is to write a narrative about how the layers of the site got there. This is the story about how people lived there and what events they experienced. In the space below, write the history of people at Rasshua 1 as you understand it from the dates you obtained and the stratigraphic sequence. Start from the bottom and explain how each layer formed and what are its chronological relationships with other layers. (In other lessons you learned or will learn about the artifacts and the food remains found at this site and will be able to understand the everyday lives of the people here better.
The last step in the process of archaeological analysis of a site is to write a narrative about how the layers of the site got there. This is the story about how people lived there and what events they experienced. In the space below, write the history of people at Rasshua 1 as you understand it from the dates you obtained and the stratigraphic sequence. Start from the bottom and explain how each layer formed and what are its chronological relationships with other layers. (In other lessons you learned or will learn about the artifacts and the food remains found at this site and will be able to understand the everyday lives of the people here better.
Rasshua 1 Test Pit 1 Profile